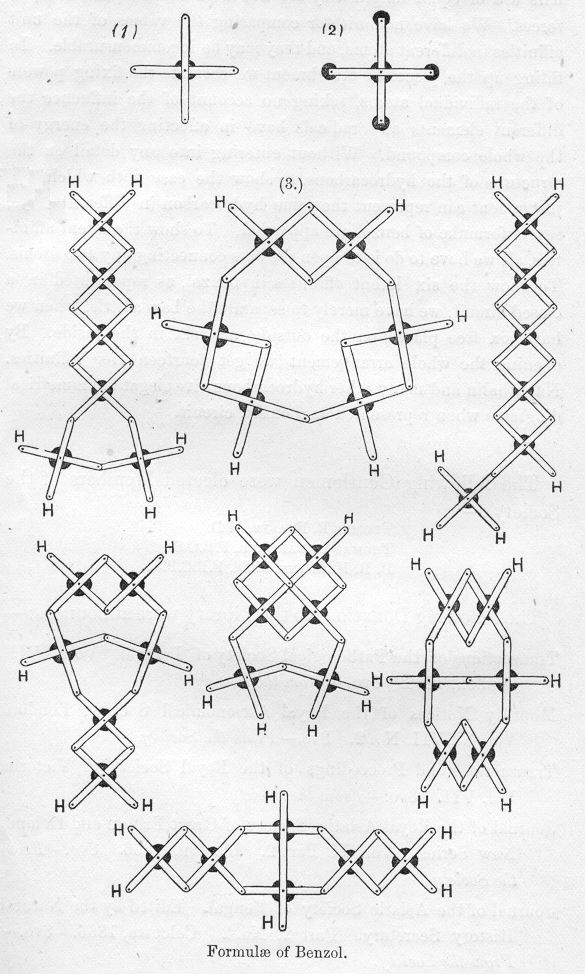
Mechanical arrangement adapted to illustrate structure in the non-saturated hydrocarbons by the chemist James Dewar. The model is made of bars, clamped together so as to allow free motion. In order to make the combination look like an atom, a thin round disc of blackened brass can be placed under the central nut. At the ends of the arms are holes to connect one carbon atom with another by means of a nut.
The structure at the bottom right of his figure is now called "Dewar benzene".
I bring before the Society a simple mechanical arrangement adapted to illustrate structure in the non-saturated hydrocarbons. This little device is the mechanical representative of Dr. C. Brown's well-known graphic notation. A series of narrow thin bars of brass of equal length are taken, and every two of the bars clamped in the centre by a nut, so as to admit of free motion the one on the other. Such a combination represents a single carbon atom with its four places of attachment. In order to make the combination look like an atom, a thin round disc of blackened brass can be placed under the central nut. At the ends of the arms are holes to connect one carbon atom with another by means of a nut. The filling up of the places of attachment may be effected by slipping on the arms round discs of brass having a groove attached, and placing the symbol of the chemical element on the round projection. A carbon atom would then look like the following diagram.
By J. Dewar, Esq. (from Proceedings of the Royal Society of Edinburgh, VI, Session 1866-67, pp. 82-86)
Dewar sent Kekulé his brass strip molecular modeling kit in 1866, and Kekulé used it to hypothesize possible molecular structures for compunds. In 1867, Kekulé used Dewar's models to come up with a reasonable (by today's standards) structure for mesitylene (1,3,5-trimethylbenzene, C9H12). Note that in the drawing, little dots appear in in the middle of some of the bonds. They appear because the drawings are of a real model, and the various atoms were screwed together at the dots. Also note that the acetone molecules in the model appear not to be symmetrical, with one C-C bond bent and the other one straight. However, the bent bond is only an illusion created by the flatness of the paper; in Kekuleé's 3-D model, all of the C-C bonds are straight.
James Deward is famous for his Researches in low temperature phenomena. See [3] to learn more.
Sources:
- J. Michael Mc Bride Models and Structural Diagrams in the 1860s
- J. Dewar, Esq., Proceedings of the Royal Society of Edinburgh, VI, Session 1866-67, pp. 82-86
- Today in Science History website